
Continued from here.
The two most important of the ‘neglected details’ in the theory of a gas turn out to be the finite size of the molecules and their mutual interactions. Johannes Diderik van der Waals understood this and developed a modified version of the ideal gas equation PV = nRT

In this equation there are two new parameters a and b which characterise the effect of the neglected details.
The parameter a characterises the way in which the electrical attraction between molecules – also described by Van der Waals – reduces the pressure below that which would be expected for an ideal gas. The squared-dependence on molar density, ρ, arises from two effects.
- Firstly in this simple model,the number of attractive interactions a single molecule experiences depends on the number of molecules in its vicinity and so is proportional to ρ
- Secondly, the effect on the pressure depends on the number of molecules per unit volume and so is proportional to ρ
Combining these two effects leads to an effect proportional to ρ2.
The parameter b characterises the finite volume occupied by the molecules themselves. We would expect this to be roughly equal to the molar volume of the substance when in it is in a solid or liquid state.
There’s a table of values of a and b parameters for a range of gases at the end of this article, but before we get calculating, it is worth noting that there are several really interesting things about this equation.
Interesting thing#1. As I mentioned in the previous article, the deviations from the ideal gas equation arise from the basic interactions between molecules. And so if we can measure these deviations from the ideal gas equation we can deduce the properties of molecular interactions. That’s right: precision measurements of mundane macroscopic properties of gases (e.g. temperature, pressure, density, thermal conductivity or speed of sound) can allow us to deduce the form of the interaction between individual molecules – something that we can calculate, but which is not accessible to direct measurement.
Interesting thing#2. The form of the Van der Waals equation derives from a particular choice of inter-molecular potential energy as illustrated qualitatively below.
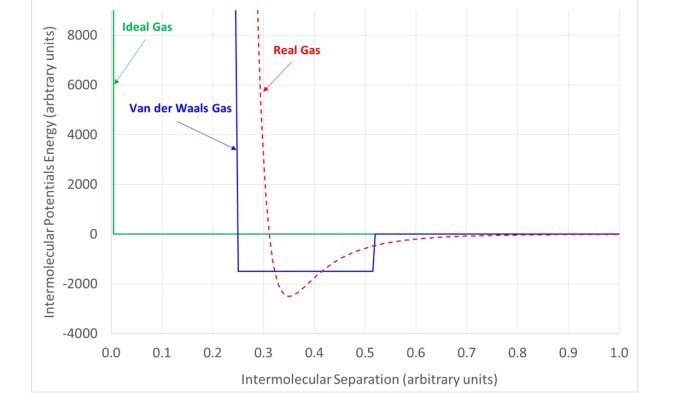
- The ideal gas equation assumes that there is no interaction between molecules and that they occupy ‘negligible volume.
- The Van der Waals equation implicitly assumes a ‘square well’ potential. This gives molecules a finite size and also implies that at close range they attract each other.
- In real gases, the inter-molecular potential retains two features of the Van der Waals interactions: a core region where molecules strongly repel other molecules and an attractive region outside this core, but the behaviour is much more subtle.
Interesting thing#3 =#2 +#1. Combining the last two observations we can now see how squeezing a gas and measuring its properties as a function of density can yield information about the pair potential.
At any instant, the molecules of a gas will have a variety of separations: some will be close together and some far apart, and the overall potential energy of the gas (assumed to be zero in the ideal gas model) will be an average over all the molecular pairs of their interaction energy. If we now squeeze the gas, we alter the average separation between molecules and hence their average potential energy. So by measuring how hard it is to compress a gas (i.e. by measuring its compressibility or bulk modulus) it is possible (after some complicated analysis) to infer the form of the pair potential between the molecules.
Interesting thing#4. Let’s get numerical. At the foot of this article is a table of values of a and b. We can use this to calculate the expected deviation from ideal gas behaviour for typical gases. After quite a lot of manipulation, it is possible to re-write the Van de Waals Equation in the following form:
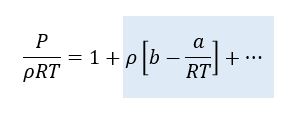
If the term on the right-hand side of the equation with the grey background were zero, then this would just describe the ideal gas equation. In this form, the term with the grey background allows is to determine the fractional error in the ideal gas equation.
Using the data for a and b in the table below I have estimated the term on the right-hand side of this equation for 20 °C (293.15 K) and atmospheric pressure (~105 Pa). The data is shown in the right-hand column of the table and amounts to typically less than 1%, even for gases composed of quite complex molecules.
Interesting thing#5. There is no shortage of interesting things to say about the Van der Waals equation, but it is late at night as I write this, so let me just add this curious note about the b parameter.
We expect the b parameter to be roughly proportional to the volume of one mole of molecules. So if we multiply this by the mass of one mole, we should get an estimate for the density of the condensed phase of the substance! Yes, that’s correct: by measuring the tiny deviations from ideal gas behaviour we can make an estimate for the density of the condensed phase (liquid or solid) of the substance!
How good are these estimates? I have highlighted water and mercury in the table below for which the density estimates are 590 kg/m3 and 11,972 kg/m3 respectively. Now these are not very close to the 1000 kg/m3 and 13,560 kg/m3 – the errors are 41% and 12% respectively. But when we consider the complexity of the structure of water I find it shocking that our estimate even comes this close.
To me this reaffirms my love of precision measurement. When we look closely at things we see all kinds of details and become aware of the connections between apparently disparate applications.
Goodnight!
================================================
Table of Van der Waals a and b constants taken from Wikipedia but using SI units. You can also download this data in spreadsheet form from the following link
| a | b | Deviation from ideal | |
| (m6 Pa/mol2) | (m3/mol) | 1 | |
| Helium | 0.00346 | 0.0000238 | 0.09% |
| Neon | 0.02135 | 0.00001709 | 0.03% |
| Hydrogen | 0.02476 | 0.00002661 | 0.07% |
| Argon | 0.1355 | 0.00003201 | -0.10% |
| Nitric oxide | 0.1358 | 0.00002789 | -0.11% |
| Nitrogen | 0.137 | 0.0000387 | -0.07% |
| Oxygen | 0.1382 | 0.00003186 | -0.10% |
| Carbon monoxide | 0.1505 | 0.00003985 | -0.09% |
| Methane | 0.2283 | 0.00004278 | -0.21% |
| Krypton | 0.2349 | 0.00003978 | -0.23% |
| Carbon dioxide | 0.364 | 0.00004267 | -0.44% |
| Hydrogen chloride | 0.3716 | 0.00004081 | -0.46% |
| Nitrous oxide | 0.3832 | 0.00004415 | -0.46% |
| Ammonia | 0.417 | 0.0000371 | -0.55% |
| Xenon | 0.425 | 0.00005105 | -0.51% |
| Silicon tetrafluoride | 0.4251 | 0.00005571 | -0.49% |
| Silane | 0.4377 | 0.00005786 | -0.50% |
| Hydrogen sulfide | 0.449 | 0.00004287 | -0.58% |
| Hydrogen bromide | 0.451 | 0.00004431 | -0.58% |
| Acetylene | 0.4516 | 0.0000522 | -0.55% |
| Fluoromethane | 0.4692 | 0.00005264 | -0.57% |
| Phosphine | 0.4692 | 0.00005156 | -0.58% |
| Hydrogen selenide | 0.5338 | 0.00004637 | -0.71% |
| Nitrogen dioxide | 0.5354 | 0.00004424 | -0.72% |
| Water | 0.5536 | 0.00003049 | -0.81% |
| Ethane | 0.5562 | 0.0000638 | -0.68% |
| Chlorine | 0.6579 | 0.00005622 | -0.88% |
| Radon | 0.6601 | 0.00006239 | -0.86% |
| Sulfur dioxide | 0.6803 | 0.00005636 | -0.91% |
| Chloromethane | 0.757 | 0.00006483 | -1.01% |
| Cyanogen | 0.7769 | 0.00006901 | -1.03% |
| Dimethyl ether | 0.818 | 0.00007246 | -1.08% |
| Mercury | 0.82 | 0.00001696 | -1.31% |
| Propane | 0.8779 | 0.00008445 | -1.13% |
| Methanol | 0.9649 | 0.00006702 | -1.35% |
| Ethylamine | 1.074 | 0.00008409 | -1.46% |
| Freon | 1.078 | 0.0000998 | -1.41% |
| Chloroethane | 1.105 | 0.00008651 | -1.51% |
| Ethanethiol | 1.139 | 0.00008098 | -1.59% |
| Carbon disulfide | 1.177 | 0.00007685 | -1.67% |
| Ethanol | 1.218 | 0.00008407 | -1.71% |
| Dimethyl sulfide | 1.304 | 0.00009213 | -1.82% |
| Butane | 1.466 | 0.0001226 | -1.97% |
| Acetone | 1.602 | 0.0001124 | -2.24% |
| Diethyl ether | 1.761 | 0.0001344 | -2.42% |
| Acetic acid | 1.771 | 0.0001065 | -2.55% |
| Acetonitrile | 1.781 | 0.0001168 | -2.52% |
| Benzene | 1.824 | 0.0001154 | -2.60% |
| Diethyl sulfide | 1.9 | 0.0001214 | -2.70% |
| Pentane | 1.926 | 0.000146 | -2.65% |
| Carbon tetrachloride | 1.97483 | 0.0001281 | -2.80% |
| Acetic anhydride | 2.0158 | 0.0001263 | -2.88% |
| Fluorobenzene | 2.019 | 0.0001286 | -2.87% |
| Ethyl acetate | 2.072 | 0.0001412 | -2.91% |
| Germanium tetrachloride | 2.29 | 0.0001485 | -3.25% |
| Cyclohexane | 2.311 | 0.0001424 | -3.31% |
| Toluene | 2.438 | 0.0001463 | -3.51% |
| Hexane | 2.471 | 0.0001735 | -3.45% |
| Chlorobenzene | 2.577 | 0.0001453 | -3.75% |
| Tin tetrachloride | 2.727 | 0.0001642 | -3.92% |
| Bromobenzene | 2.894 | 0.0001539 | -4.24% |
| Iodobenzene | 3.352 | 0.0001656 | -4.97% |